The
Climate Catastrophe
- A Spectroscopic Artifact?
by Dr. Heinz Hug
(Reactions to Dr Hug's controversial paper (downloadable zip file) here)
![]()
![]() Summary
Summary ![]()
Laboratory measurements of the infrared absorption of carbon dioxide using an FT-IR spectrometre suggest that the radiative forcing for CO2 doubling must be much less than assumed by climate scientists until now. A reduction factor of 80 is likely.
![]() Introduction
Introduction
Originally the CO2 greenhouse effect obviously was considered from the 'normal' infrared (IR) absorption [1]. Supposed with today's concentration it would exist a decadic extinction of E = 4 which means the transmission T = I/Io = 10-E has been reduced to 0.0001 (in the vicinity of the maximum peak of 15 µm). Every scientisct who is familiar with basic IR spectroscopy from analytic chemistry would agree that there is a noticeable temperature increase for a CO2 doubling. But indeed today's transmission is less by several magnitudes. According to Jack Barrett [2] the possible maximum greenhouse effect occurs within a 100 m layer near ground.
In the literature we often see emission spectra [3] and sometimes absorption spectra of CO2 as well [4]. But an interested chemist is missing molar related figures of the extinction. CO2 is one of the most intensively investigated molecule. This is why we find enough quantitative data in the literature [5] and of course, there are the HITRAN spectra. But unfortunately there are quite different opinions within the scientific community. Moreover the warming projections are continuously reduced. This is why own measurements were carried out [6].
![]() Metering
method
Metering
method
A 10 cm glass cylinder (150 cm3, with IR-transparent window) was filled with synthetic CO2-free and vapour-free air. Then a microlitre syringe was used to add CO2 so that the concentration was 357 ppm (concentration in 1993). Moreover 2.6% water vapour was added. Applying the IR beam source (a so-called Globar , an electrically heated silicon carbide bar at 1000 to 1200 degC and an adjustable interference filter) on one side, the absorption spectrum arriving at the other end was recorded. Then CO2 was added to make 714 ppm. The equipment was an FTIR spectrometer "Bruker IFS 48" coupled to a PC. The program OPUS was used as analyzing software. A zero bias measurement was made to be subtracted later.
![]() Results
Results
Fig. 1 shows the unprocessed spectrum of the 15 µm band for 357 ppm CO2 and 2.6% H2O.

Fig. 1: Unprocessed spectrum of the 15 µm
band
The absorption peak depends on the spectral resolution which was 2/cm for this spectrometer. With a finer resolution, e.g. 0.5/cm, the peak would become higher and sharper, thus yielding a higher extinction coefficient. The R- (DeltaJ = +1) and the P- (DeltaJ = -1) can be clearly identified as well as the Q-branch (DeltaJ = +0) of the n3 band (15 µm or 667 cm-1). The n2 band (4.2 µm or 2349 cm-1) which only has an R- and P-branch, was measured as well. The decadic extinction coefficients at the band maximum were evaluated as
e = 29.9 m2/mol for n2 and e = 20.2 m2/mol for n3
To calculate the transmission in the total atmosphere, an average CO2 content was taken (from the volume of the atmosphere and the mass) as c = 1.03*10-3 mol/m3. Inserting the above molar extinction, the value for c and the homosphere layer thickness (h = 105 m) into Lambert-Beer's law, yielding a decadic extinction
E(n2) = 29.9 m2/mol * 1.03 * 10-3 mol/m3 * 105 m = 3080
In the same way we find E(n3) = 2080. This means that the transmission T around the peak maxima, defined as 10-E, amounts for 357 ppm to
T(n2) = 10 -3080 and T(n3) = 10 -2080
These are extremely small transmission values which are making any greenhouse increment by CO2 doubling absolutely impossible. Jack Barrett found similar results [2] using spectroscopic and kinetic considerations - tapping into a vasp nest and creating a still vivid discussion [7 - 10].
Inserting e = 20.2 m2/mol for the n3 band into Lambert-Beer's law, using 357 ppm for the CO2 concentration and a 10 m layer, we find the extinction
E = 20.2 m2/mol * 0.0159 mol/m3 * 10 m = 3.21
As the transmission T = 10-3.21 is 0.6 per mille, we conclude that the relative absorption around the peak is 1-T = 99.94% which takes place already within a 10 m layer near ground. Of course, here only the absorption of an IR beam has been accounted for - but shouldn't we expect that the 15 µm band is re-radiated by CO2 molecules? According to Jack Barrett [2], most of this energy is thermalized from collisions with other molecules (N2, O2, H2O) long before re-radiation takes place so that the energy gets out of the CO2 band. Half of this thermalized IR can be considered to enhance the back-radiation and thus warming the ground.
The conclusion of total radiative saturation was tried to be cancelled out sophistically by assuming that (more) tropospheric warming may be caused by radiative transfer in a cascade model with sequential IR absorption and re-emission by CO2. Then - only within the absorption band - the radiative transfer eqation has to be solved [11] with dz being a small distance, sa the absorption coefficient and n the number of molecules:
dl = l*n*sa*dz - B*n*sa*dz
This results in a complex equation system [12]. One problem is that the radiative transport depends on the temperature gradient in the atmosphere - but this cannot be preset but ought to be calculated. So an iterative scheme has to be applied. The modeller then has to determine to what fraction the soil is warmed by back-scattered IR and by warmed air.
For the edges of the 15 µm band the absorption area is certainly smaller than what is used by IPCC. IPCC [1990, p. 48] states "The effect of added carbon dioxide molecules is, however, significant at the edges of the 15 µm band, and in particular around 13.7 and 16 µm" [13].
To check for this we hyothesized that we can omit the decadic extinction increase above E = 3 (or T = 10-3) for CO2 doubling. For this case we took the total integral of the bands till the ends of the R- and P-branch with E = 0 for the computed transmission spectrum of the whole atmosphere prepared from digitally recorded spectral measurements. Fig 2 shows in principle - not in correct proportion - how the band edges were integrated.
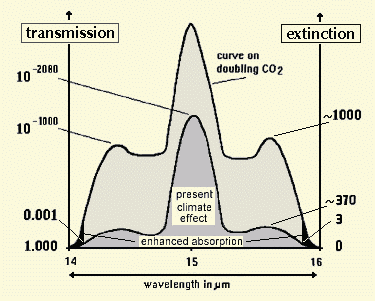
Fig. 2: Spectral processing scheme
We integrated from a value E = 3 (above which absorption deems negligible, related to the way through the whole troposphere) until the ends (E = 0) of the R- and P-branch. So the edges are fully considered. They start at 14.00 µm for the P-branch and at 15.80 µm for the R-branch, going down to the base line E=0. IPCC starts with 13.7 and 16 µm [13]. For the 15 µm band our result was:
| 15 µm band | 357 ppm | 714 ppm |
| total integral 624,04 cm-1 - 703,84 cm-1 |
0.5171/cm | 1.4678/cm |
| sum of slope integrals | 1.11*10-4/cm | 9.79*10-4/cm |
Table 1: 15 µm band total integration and slope integrals E = 0 to E = 3
Crucial is the relative increment of greenhouse effect . This is equal to the difference between the sum of slope integrals for 714 and 357 ppm, related to the total integral for 357 ppm. Considering the n3 band alone (as IPCC does) we get
(9.79*10-4 cm-1 - 1.11*10-4 cm-1) / 0.5171 cm-1 = 0.17 %
![]() Conclusions
Conclusions
It is hardly to be expected that for CO2 doubling an increment of IR absorption at the 15 µm edges by 0.17% can cause any significant global warming or even a climate catastrophe.
The radiative forcing for doubling can be calculated by using this figure. If we allocate an absorption of 32 W/m2 [14] over 180º steradiant to the total integral (area) of the n3 band as observed from satellite measurements (Hanel et al., 1971) and applied to a standard atmosphere, and take an increment of 0.17%, the absorption is 0.054 W/m2 - and not 4.3 W/m2.
This is roughly 80 times less than IPCC's radiative forcing.
If we allocate 7.2 degC as greenhouse effect for the present CO2 (as asserted by Kondratjew and Moskalenko in J.T. Houghton's book The Global Climate [14]), the doubling effect should be 0.17% which is 0.012 degC only. If we take 1/80 of the 1.2 degC that result from Stefan-Boltzmann's law with a radiative forcing of 4.3 W/m2, we get a similar value of 0.015 degC.
Kondratjew and Moskalenko are referring to their own work [15] - but when we checked their Russian book on that page, it turned out that this was nothing but an index of terms and nowhere else a deduction of this broadly referred 7.2 K figure [16] could be found. It should be mentioned that the radiative forcing for the present CO2 concentration varies considerably among references. K.P. Shine [17] specifies a value of 12 K whereas according to R. Lindzen CO2 only accounts for about 5% of the natural 33 degC greenhouse effect. This 1.65 degC is less than a quarter of the value used by IPCC and leads to a doubling sensitivity of 0.3 to 0.5 degC only [18].
What is really true? Is there anybody to present a scientific derivation or a reference where this figure is not copied or just stated from assumptions, but properly calculated?
![]() References
References
[1] Roger Revelle, Scientific American, 247, No.2, Aug. 1982, 33 - 41
[2] Jack Barrett, Spectrochim. Acta Part A, 51, 415 (1995)
[3] R.A. Hanel et al. Journal of Geophysical Research, 77, 2629 - 2641 (1972)
[4] Hermann Flohn, Nachr. Chem.Tech.Lab, 32, 305-309 (1984)
[5] L.S.Rothman et al., Appl.Opt. 26, 4058 (1987)
[6] Heinz Hug, Chemische Rundschau, 20. Febr., p. 9 (1998) and: Klima 2000 (Heuseler), 2, 23-26 (1998) 1/2 and: http://www.wuerzburg.de/mm-physik/klima/artefact.htm
[7] Paul S. Braterman, Spectrochim. Acta Part A, 52, 1565 (1996)
[8] Keith Shine, Spectrochim. Acta Part A, 51, 1393 (1995)
[9] John Houghton, Spectrochim. Acta Part A, 51, 1391 (1995)
[10] Richard S. Courtney, Spectrochim. Acta Part A, 53, 1601 (1997)
[11] Richard P. Wayne, Chemistry of Atmospheres, Oxford University Press, 2nd. Edition, 44-49 (1991),
[12] Murry L. Salby, Fundamentals of Atmospheric Physics, Academic Press, 198-257 (1996)
[13] Climate Change 1990. The IPCC Scientific Assessment, p. 49
[14] K.Ya. Kondratyev,N.I. Moskalenko in J.T.Houghton, The Global Climate", Cambridge Universitiy Press, 225-233 (1984)
[15] K.Ya. Kondratyev, N.I. Moskalenko, Thermal Emission of Planets, Gidrometeoizdat, 263 pp (1977) (in Russian)
[16] C.-D. Schönwiese, Klimaänderungen, Springer-Verlag Berlin Heidelberg, p. 135 (1995)
[17] Henry Charnock, Keith P. Shine, Physics Today, Dec 1993, p. 66
[18] Richard S. Lindzen, Proc. Nat. Acad. of Sciences, 94, 8335-8342 (1997) 8 and (in German) Klima 2000 (Heuseler), 2, 3-8 (1998) 5/6
July 31, 1998
Heinz Hug, Wiesbaden, Germany
Fax +49/611-543301
http://www.john-daly.com//artifact.htm
Return to Climate Debate Guests Page
Return to "Still Waiting For Greenhouse" main page